News Release
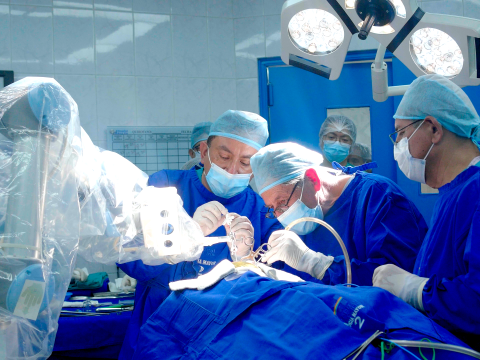
Brain Navi Biotechnology has officially received U.S. FDA 510(k) clearance for its neurosurgical robot NaoTrac, now classified as a "Stereotaxic Guiding Surgical Device." Following earlier approvals from Taiwan’s TFDA and the European CE Mark, this milestone reinforces NaoTrac’s international recognition in safety and clinical effectiveness. Designed to assist with various neurosurgical procedures, NaoTrac integrates AI-powered autonomous navigation and robotic precision to support safer, more efficient surgeries in the operating room.
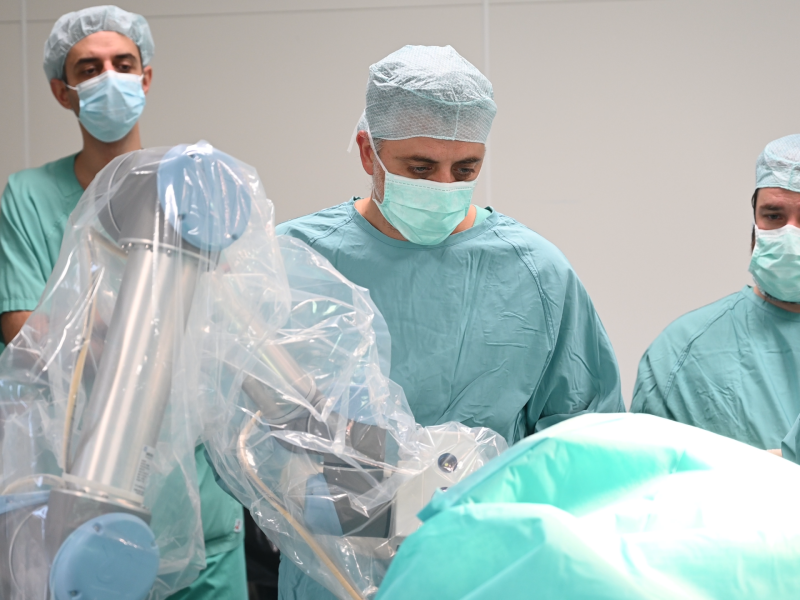
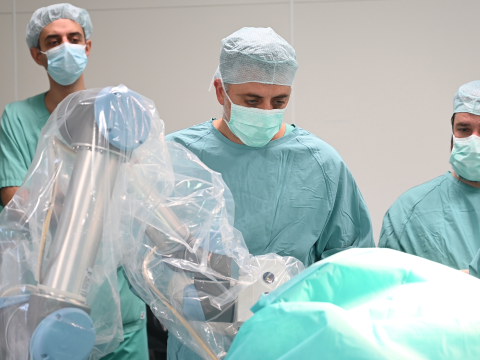
Cottbus, Germany – The first surgeries with the neurosurgical navigation robot NaoTrac have now been successfully started at the Medizinische Universität Lausitz – Carl Thiem.
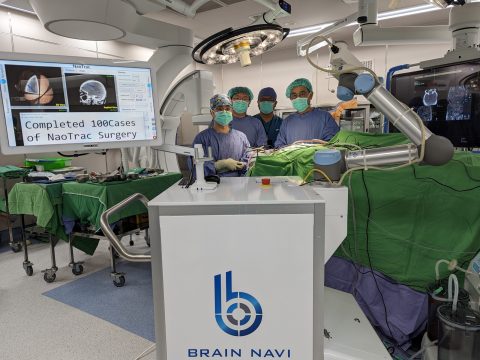
NaoTrac from Brain Navi completed 100 clinical cases in An-Nan National Hospital.
News Release
Cottbus, Germany – The first surgeries with the neurosurgical navigation robot NaoTrac have now been successfully started at the Medizinische Universität Lausitz – Carl Thiem.
Brain Navi Biotechnology has officially received U.S. FDA 510(k) clearance for its neurosurgical robot NaoTrac, now classified as a "Stereotaxic Guiding Surgical Device." Following earlier approvals from Taiwan’s TFDA and the European CE Mark, this milestone reinforces NaoTrac’s international recognition in safety and clinical effectiveness. Designed to assist with various neurosurgical procedures, NaoTrac integrates AI-powered autonomous navigation and robotic precision to support safer, more efficient surgeries in the operating room.
NaoTrac from Brain Navi completed 100 clinical cases in An-Nan National Hospital.
Brain Navi announced to join CNS in Washington D.C. and EANS 2023 Barcelona, Spain to demonstrate Naotrac and KrystoLens
Bolivia and Taiwan online webinar discuss about "The Future of Robotic Surgery"
No more posts to show